Halle Magnetic Resonance Center – HallMaRC
MLU’s Faculty of Natural Sciences II hosted three research groups that specialized on magnetic resonance spectroscopy, which is among the most powerful and versatile classes of methods for the investigation of structure and dynamics of polymers at molecular (atomic) scale. Our unique equipment, partially situated in a special custom-built facility, comprised of 8 superconducting magnets for high-field nuclear and electron spin resonance spectroscopy, plus a number of commercial and custom-built low-field instruments. Synthetic and biological macromolecules were the central focus of our research activities.
In magnetic resonance, one uses the phenomenon that the energy levels of spin magnetic moments of specific nuclear isotopes or unpaired electrons become non-degenerate once they are placed in a strong magnetic field. The energy differences between these energy levels of detectable individual spins are in the radio-frequency (nuclear spins) or microwave frequency (electron spins) range. They are modulated by details of the local surroundings and thus report about the molecular structure and dynamics. We make use of this unique capability of nuclear magnetic resonance (NMR) or electron paramagnetic/ spin resonance (EPR or ESR) in a large number of independent and collaborative projects, most of which evolve around questions in the wider field of soft matter sciences.
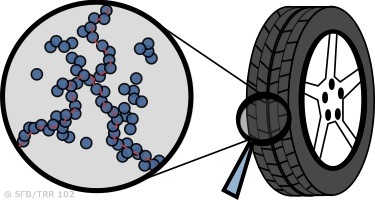
Work in Kay Saalwächter’s group mainly focuses on NMR applications to synthetic soft matter such as polymers or liquid crystals. Of particular relevance for applications in everyday life are the studies of chain dynamics in elastomers, i.e., polymer networks, as for instance used in car tire applications. The molecular fluctuations in such materials depend on the density of crosslinks between the individual chains, which in turn determine the mechanical properties. We have optimized simple low-field proton NMR spectrometers for the detection of inhomogeneities in such elastomers, and use this new and unique probe to establish quantitative structure-property relations. On top of this, nanoparticles are often needed to optimize the mechanical performance of elastomeric materials. Yet, the influence of the particle surface on the dynamics of the surrounding polymer can be large and is not well understood. Again, NMR can be used to quantify such effects, and thus to elucidate the role of “nano-confinement” on polymer dynamics.
 Dariush Hinderberger, workgroup Complex Self-Organizing Systems[/caption]
Dariush Hinderberger, workgroup Complex Self-Organizing Systems[/caption]
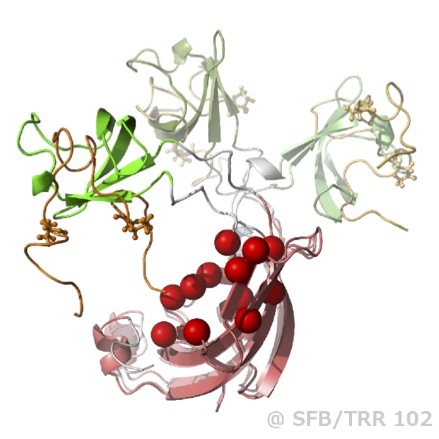
Biological and synthetic soft matter has a remarkable ability to self-assemble into larger, complex and even functional structures solely using non-covalent interactions (e.g. electrostatic and hydrophobic forces). Dariush Hinderberger’s research group uses EPR spectroscopy as its main research tool to study soft matter from the underlying fundamental interactions to more applied questions. Research in the group currently has a focus on solvation and self-assembly of small molecules (e.g. in aqueous solutions or ionic liquids), on complex biomedical questions (e.g. abundant transport proteins such as albumin or intrinsically disordered proteins, IDPs), and applications such as understanding interactions and dynamics of transported molecules in thermoresponsive polymers for drug delivery.